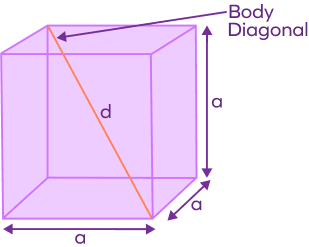
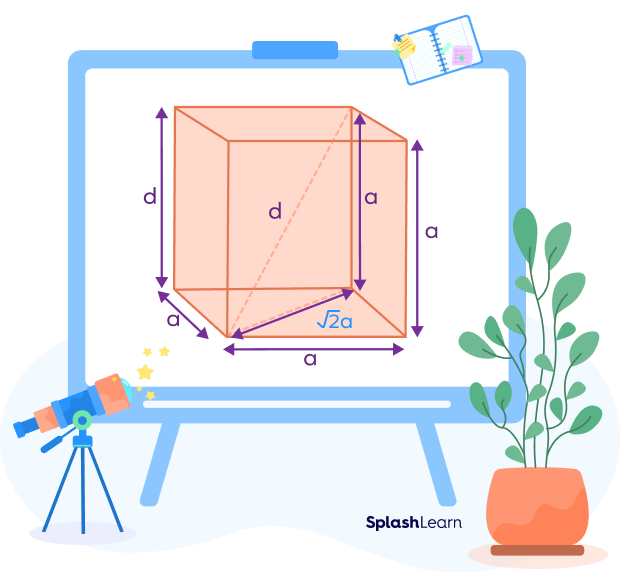
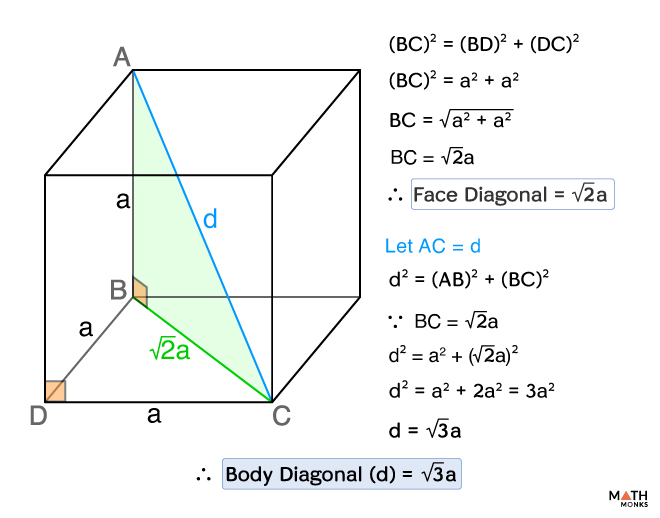
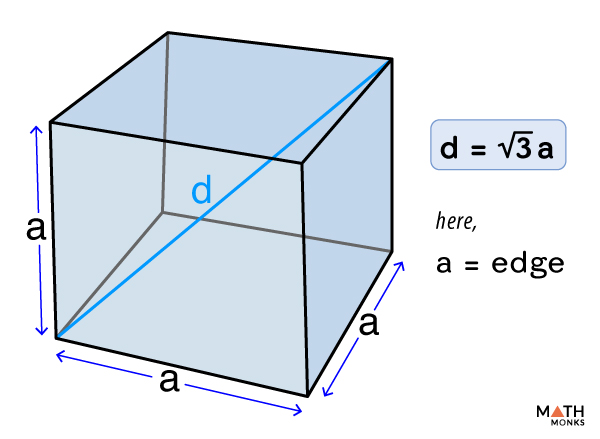
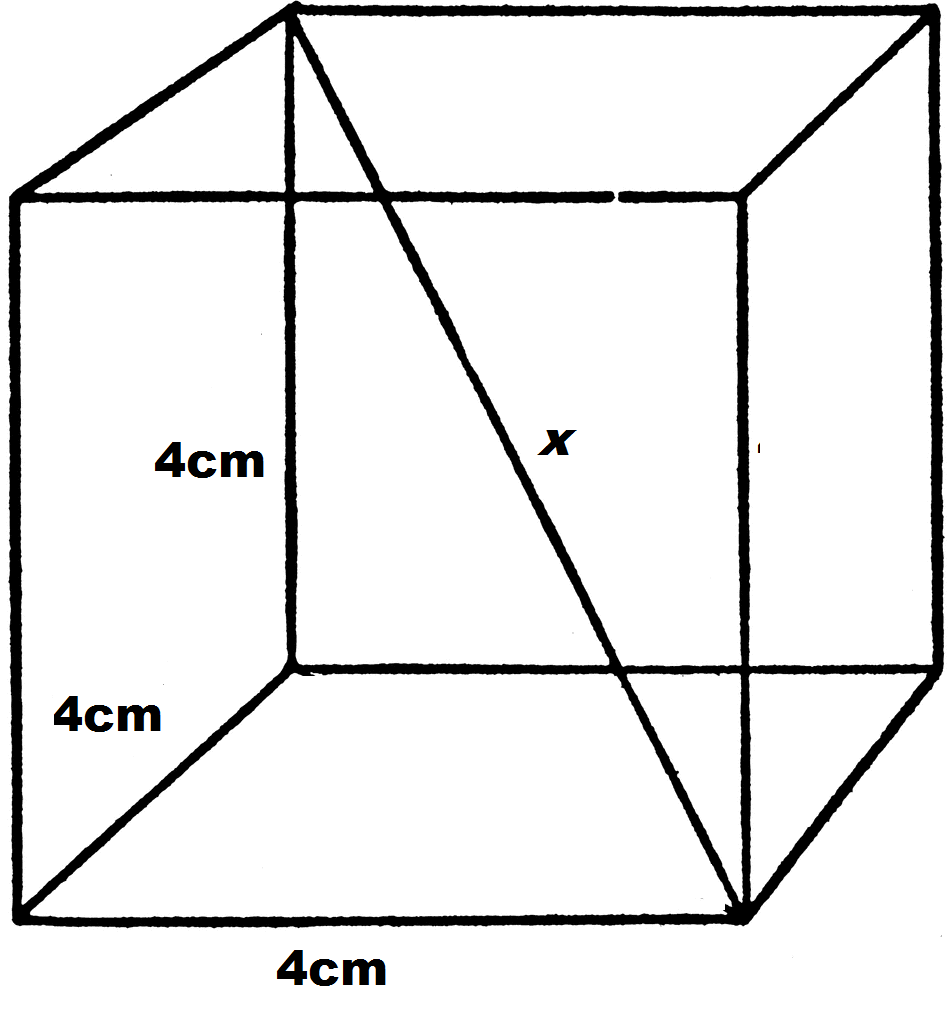
![If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$ If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$](https://www.vedantu.com/question-sets/720721fd-3ab5-4f3a-8864-68a0dfdd06901210945075913440462.png)
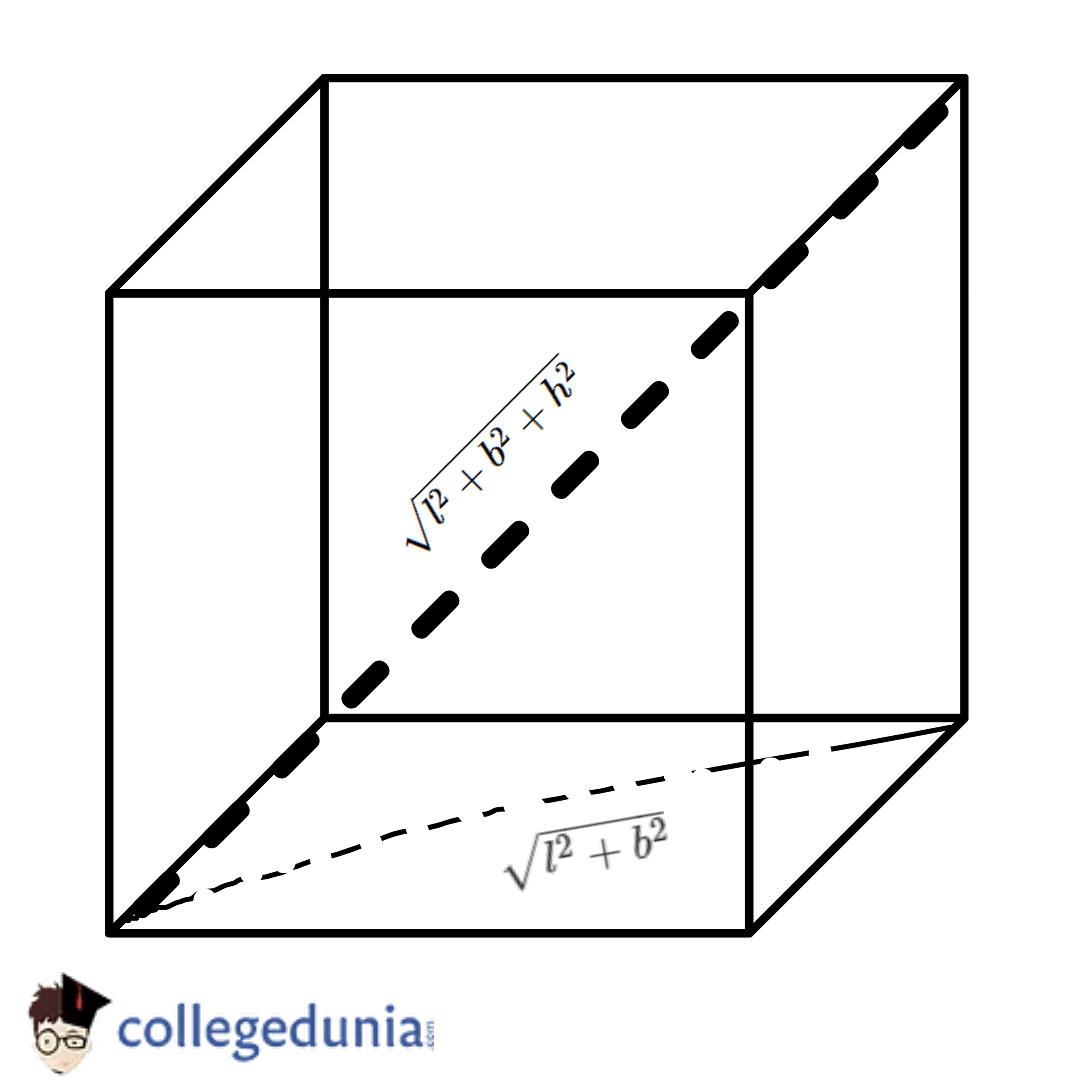
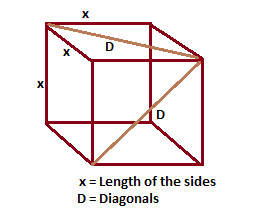
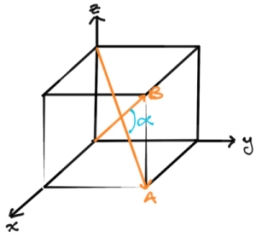
If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$
Why do we consider the diagonal of a cube in the calculation of the edge length of a BCC crystal? - Quora

13. If the body diagonal of cube crystal is 2.50 A, the approximate edge length of unit cell is (1) 2 A (2) V2 A (3) 3 A (4) 13 A D i n structure and